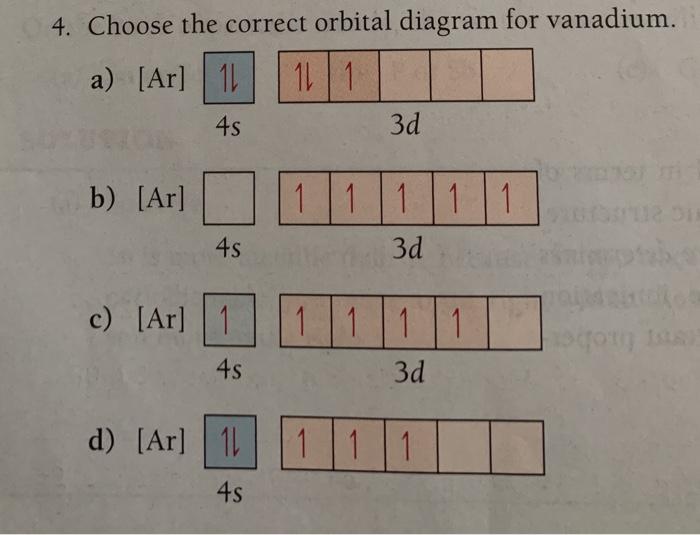
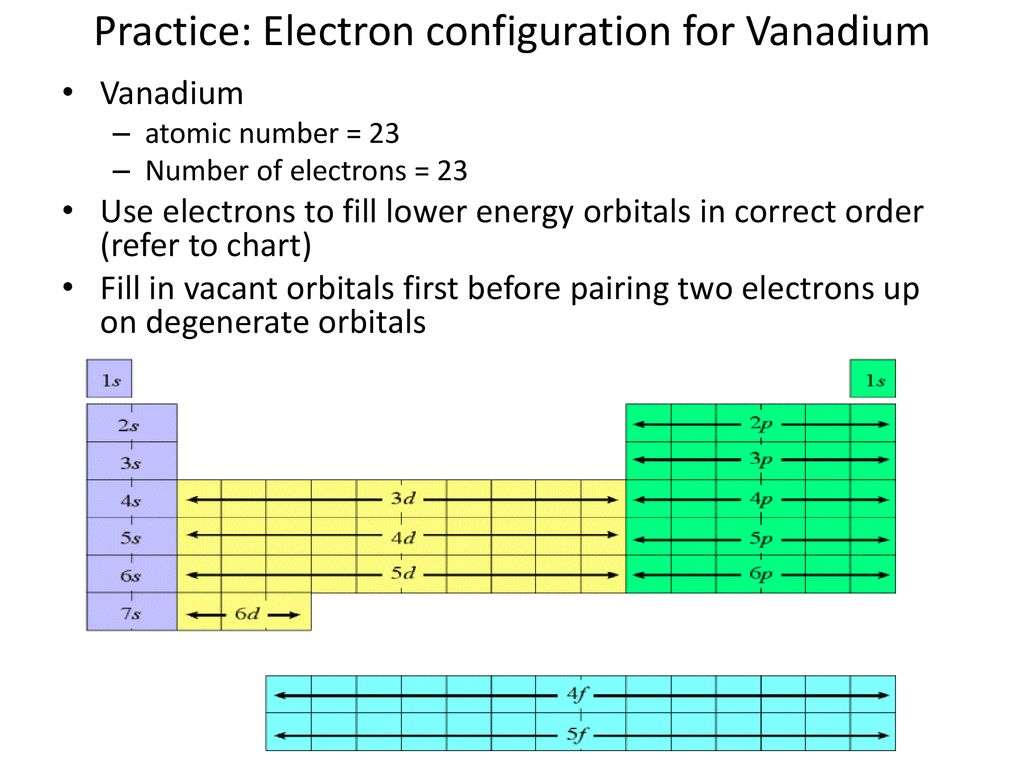
44 choose the correct orbital diagram for vanadium.
Doesn't the 5s come before the 4d? I chose [Kr]5s^2 on a quiz but it was wrong. Therefore, the ground state electron configuration for Zr 2+ is : [Kr]4d 2 5s 2. In^+1 [Kr] 5s2 4d10. 1s2, 2s2, 2p6. 1) nuclear charge and relative energy of 3d and 4s orbitals, 2) relative e-e repulsions in 3d and 4s orbitals, 3) exchange energy. Booster Classes. Need an editable periodic table to edit? Enter the ... The Electron Configuration. Q. The innermost electron shell of an atom can hold up to _ electrons. Solved • Mar 17, 2021. The Electron Configuration. Q. The element that corresponds to the electron configuration 1s22s22p63s23p64s13d5 is: A) titanium B) vanadiumC) mangneseD) chromium. Solved • Jan 27, 2021.
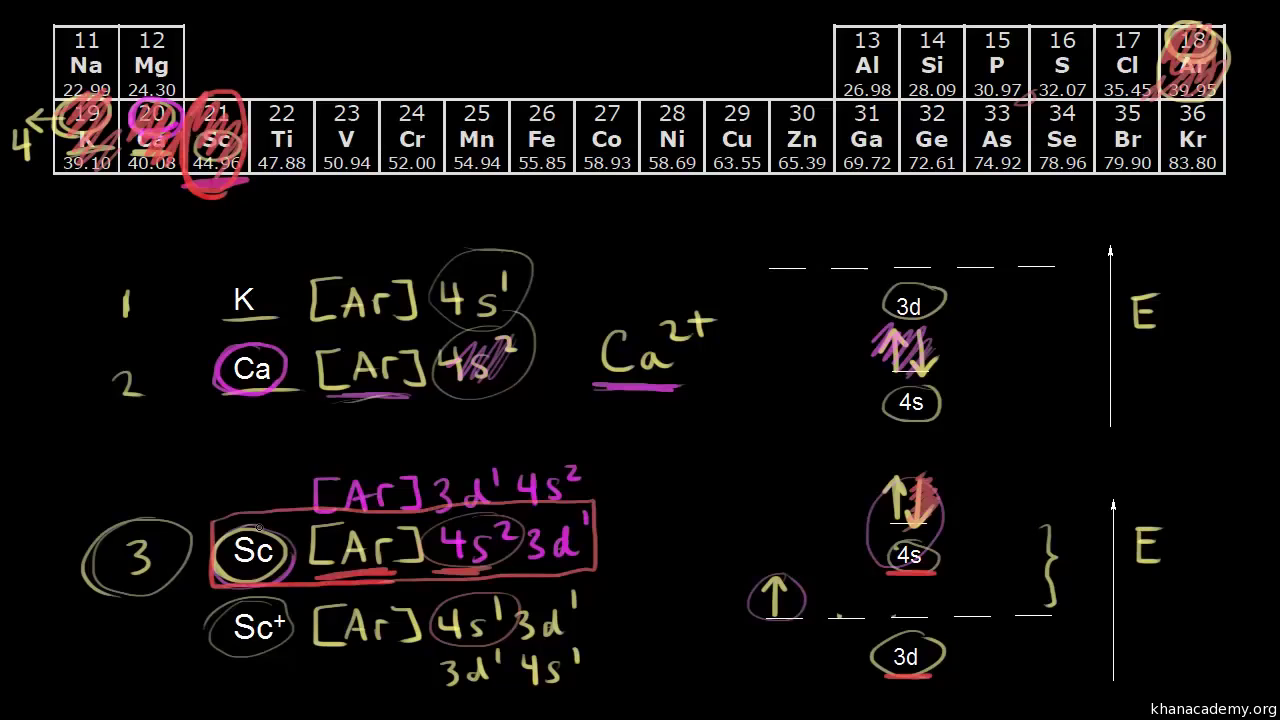
The orbital diagram in which 'aufbau principle' is violated, is represented by the option (B). Aufbau principle: In the ground state of the atoms, the orbitals are filled with electrons in order of increasing energy. The 2s sub-energy level is at a lower energy than the 2p sub-energy level. For 2s, n=2 and l=0 .

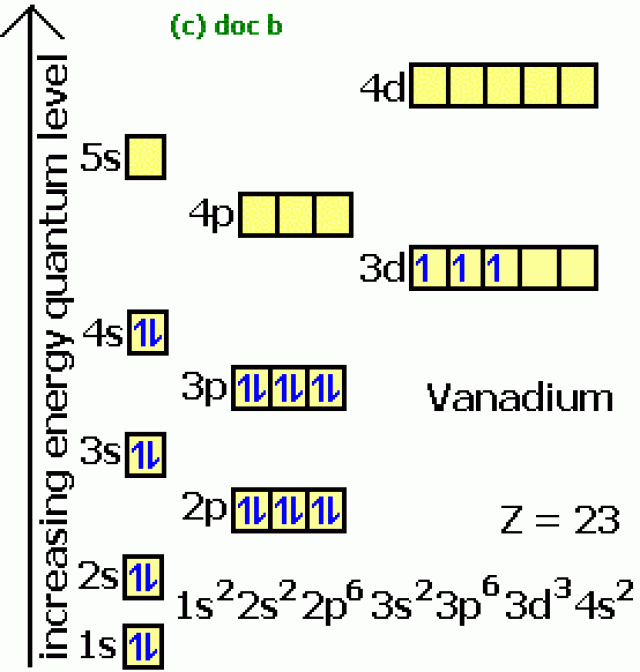
Choose the correct orbital diagram for vanadium.
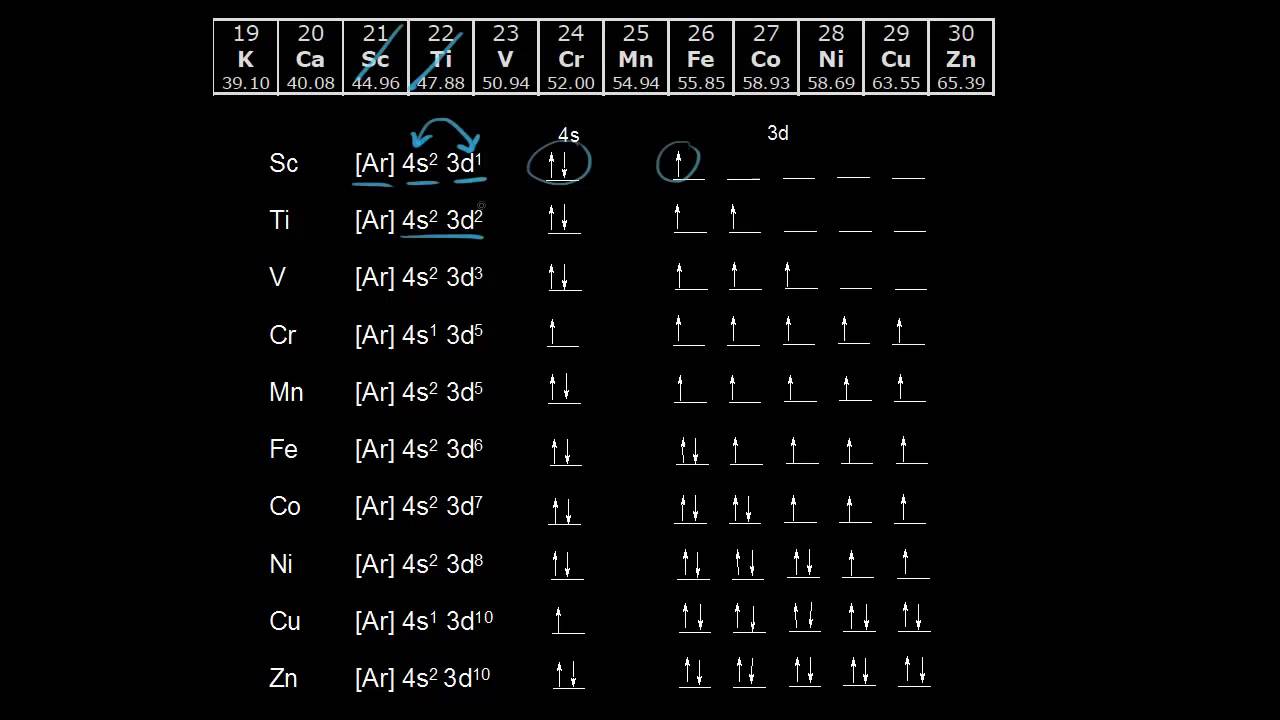
Cr and Cu, as well as Cu and Ag, are exceptions in the "typical" filling order.Orbital Diagram. 1s Vanadium pentoxide (V2O5) is used as a catalyst, dye and color-fixer. Sources Found in the minerals patronite (VS4), vanadinite [Pb5 (VO4)3Cl], and carnotite [K2 (UO2)2 (VO4)H2O]. Pure metal produced by heating with C and Cl to produce VCl3 which is . Give the complete electronic configuration for Mn. Choose the statement that is TRUE. *Core electrons effectively shield outer electrons from nuclear charge. *Core electrons are the easiest of all electrons to remove. *Outer electrons efficiently shield one another from nuclear charge. *Valence electrons are most difficult of all electrons to ... Choose the correct orbital diagram for vanadium [AR] 4s [||] [ { || } { | } { } { } { } ] ... metallic character decreases as you move to the right across a row in the periodic table and decreases as you move down a column d) metallic character increases as you move to the right across a row in the periodic table and decreases as you move down ...
Choose the correct orbital diagram for vanadium.. After the 4s is full we put the remaining six electrons in the 3d orbital and end with 3d6. Therefore the Iron electron configuration will be 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 6. Note that when writing the electron configuration for an atom like Fe, the 3d is usually written before the 4s. Both of the configurations have the correct numbers of ... What is the Orbital Diagram For Nitrogen? When we talk about the orbital diagram, we first need to understand what exactly it means. Therefore, during exams, the student can expect questions related to this topic so it is important that the students must go through it. If you are new to such a subject and looking for periodic tables and their ... Orbital Diagram. 1s ... Vanadium pentoxide (V2O5) is used as a catalyst, dye and color-fixer. Sources Found in the minerals patronite (VS4), vanadinite [Pb5(VO4)3Cl], and carnotite [K2(UO2)2(VO4)2.3H2O]. Pure metal produced by heating with C and Cl to produce VCl3 which is heated with Mg in Ar atmosphere. How to choose the correct orbital diagram for vanadium? Problem 4SAQ: Choose the correct orbital diagram for vanadium. step-by-step solutions; Solved by professors &. Oxidation States, +5,2,3,4. Electrons Per Shell, 2 8 11 2. Electron Configuration, [ Ar] 3d3 4s2. If playback doesn't begin shortly, try restarting your device. Which is the ...
Identify the choice that best completes the statement or answers the question. ... Vanadium (V) ... What is the correct orbital diagram for Ca2+? Choose the one you consider correct and record your choice in soft pencil on the separate Answer Sheet. ... D End-to-end orbital overlap results in a bond with electron density above and below the bond ... 9 The diagram represents the energy changes for some reactions. Orbital Diagram For Vanadium (V) | Vanadium Electron Configuration. February 18, 2021 by Sneha Leave a Comment. Vanadium Electron Configuration: When it comes to electronic configuration, it is one of the major topics in chemistry as we have mentioned before in our article. And in Cr and Cu the excites 1 electrons because by doing that both 4s and 3d orbital gains half filled i.e the stable configuration. So in vanadium if u excite 1 electron the d orbital will still not be half filled. But 4s orbital will be half filled. Since fully filled orbital are more stable than half filled the vanadium will have 2 ...
It is mixed with other metals to make very strong and durable alloys. Vanadium pentoxide (V2O5) is used as a catalyst, dye and color-fixer. Atomic Number 23 Learn more about the atomic number. Description Soft, ductile, silvery-white metal. Resistant to corrosion by moisture, air and most acids and alkalis at room temperature. Atomic Mass 50,9415 Electron Configuration Chart for All Elements in the Periodic Table. To write the configuration for the Nickel ions, first we need to write the electron configuration for just Nickel (Ni). A representation of the atomic spectrum of zirconium. Uncategorized Choose the correct orbital diagram for vanadium. Call For Cell Phone Repair (613) 779-8812. Transcribed image text: 4. Choose the correct orbital diagram for vanadium. a) [Ar] 1L 45 3d b) [Ar] 1 1 1 1 1 3d 4s c) [Ar] 1 1 4s 3d d) [Ar] 11 4s. The diagram (not to scale) summarises the energies of the orbitals up to the 4p level. The oddity is the position of the 3d orbitals. They are shown at a slightly higher level than the 4s - and so it is the 4s orbital which will fill first, followed by all the 3d orbitals and then the 4p orbitals.
Transcribed image text: Using crystal field theory, draw an electron box energy level diagram for the valence d orbitals on the nickel atom in a tetrahedral | NiC14 |-complex. Your diagram should show the relative energy of each orbital, and the number of electrons in each orbital Note: to make your diagram easier to read, label the d orbitals with just the subscript.
Refer to the explanation. The electron configuration of manganese, atomic number 25, is "1s"^2"2"^2"2p"^6"3s"^2"3p"^6"3d"^5"4s"^2". The diagram below represents the electron configuration as an orbital diagram.
The 4s electrons are lost before the 3d electrons are lost. Reason: All orbitals shrink when the atom becomes a cation; electron-electron repulsion would soar in a shrunken 4s orbital. The electron configuration of Fe2+ is [Ar] 3d6. The next electron is removed from the 3d subshell. The electron configuration of Fe3+ is [Ar] 3d5.
Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table.. The electron configuration for the first 10 elements. H #1s^1# He #1s^2# Li #1s^2 2s^1# Be #1s^2 2s^2# B #1s^2 2s^2 2p^1# C #1s^2 2s^2 2p^2# N #1s^2 2s^2 2p^3# O #1s^2 2s^2 2p^4# F #1s^2 2s^2 2p^5#
C.) An electron in the 3p orbital. Choose the correct electron configuration for Se. A.) 1s2 2s2 2p6 3s2 3p4 B.) 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4 C.) 1s2 2s2 2p6 3s2 3p6 4s2 4p4 D.) 1s2 2s2 2p6 3s2 3p6 4s2 3d4. B.) 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4. What is the orbital diagram for Vanadium? [Ar] 4s= 1 up 1 down. Next orbital: 3 up arrows
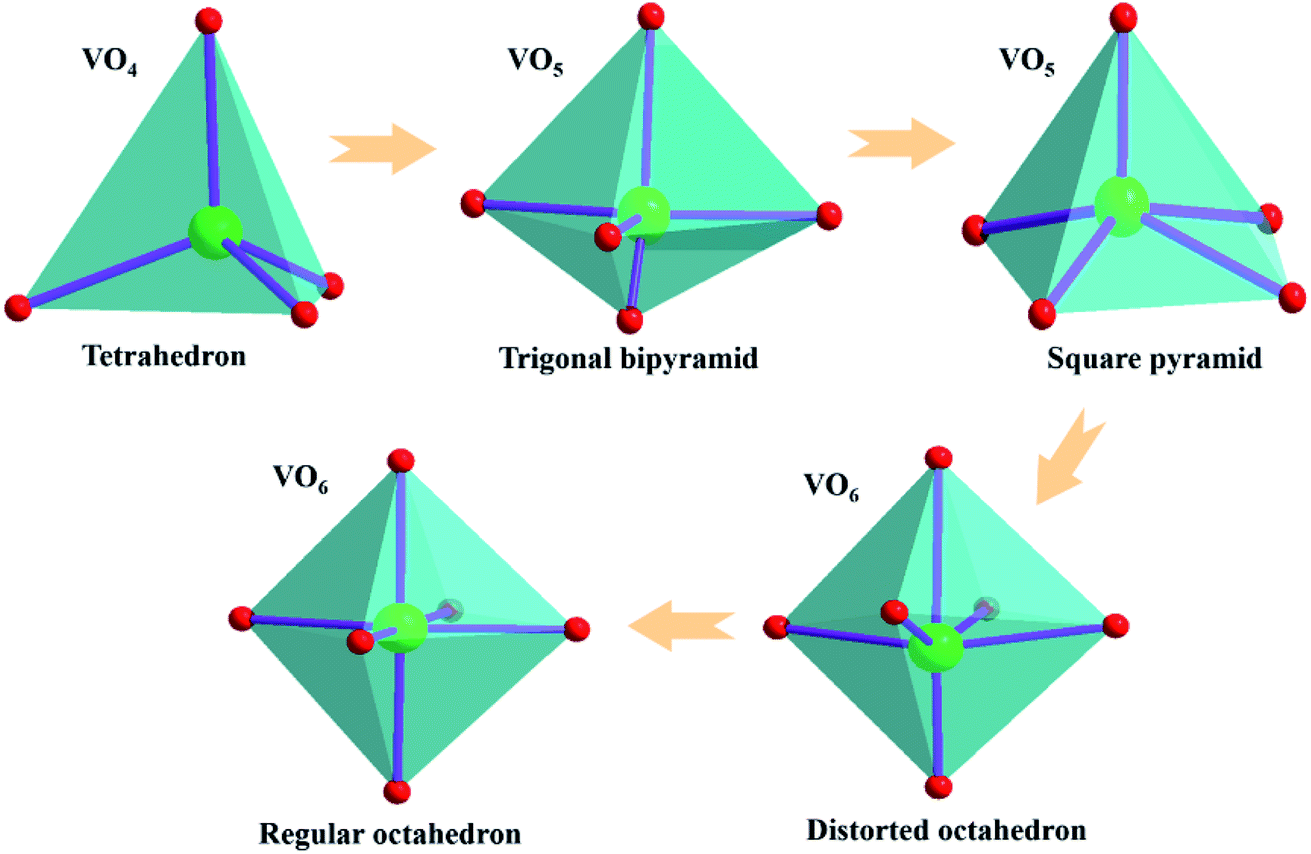
Vanadium Based Cathodes For Aqueous Zinc Ion Batteries From Crystal Structures Diffusion Channels To Storage Mechanisms Journal Of Materials Chemistry A Rsc Publishing
Which is the correct orbital diagram for vanadium? ... Choose the element with the more negative (more exothermic) electron affinity from each pair.
Orbital diagrams are pictorial descriptions of the electrons in an atom. Orbital Diagram Ar Wiring Diagrams The pauli exclusion principle says that only two electrons can fit into an single orbital. Fe orbital diagram. What is the electron configuration and orbital diagram of. Three rules are useful in forming orbital diagrams.
Both of the configurations have the correct numbers of electrons in each orbital, it is just a matter of how the electronic configuration notation is written (here is an explanation why). Therefore we have (still incorrect) 1s 2 2s 2 2p 6 3s 2 3p 6 3d 4 4s 2. Correct Electron Configuration for Chromium (Cr)
Problem 4SAQ: Choose the correct orbital diagram for vanadium. step-by-step solutions; Solved by professors &. Oxidation States, +5,2,3,4. Electrons Per Shell, 2 8 11 2. Electron Configuration, [ Ar] 3d3 4s2. How to Write Electron Configurations and Orbital Diagrams

Which Of The Following Choices Is The Correct Electron Configuration For A Vanadium Atom 3d A Homeworklib
Electron configuration for Vanadium (V) 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d1. Electron configuration for Yttrium (Y) Carbon (C) What element is represented by this orbital diagram? ... What element is represented by this orbital diagram? THIS SET IS OFTEN IN FOLDERS WITH... 6.2 Classifying Elements. 17 terms. mnethercott. SVHS Chem 8.1 ...
Orbital filling of the orbitals in an element takes place according to the following rules. • Electron occupies orbitals so as to minimize the energy of the atom; therefore, lower energy orbitals fill before higher energy orbitals. Orbitals fill in the following order: • Orbital can hold only up to two electrons each.
Get the detailed answer: Q4. Choose the correct orbital diagram for vanadium. 45 3d a) [Ar] 1 11 11 b) [Ar] 1|11|11 c) [Ar] 1 1111111 1) (ar ...
Answer: Go to the following website to find the orbital diagram of any element of any Oxidation state. Orbital Energy Diagram and Atomic Electron Configuration Tool As for an actual diagram (per Wiki) is above. You can also find the electron configuration on both websites. For an overview and ...
We're being asked to choose the correct orbital diagram for vanadium.For that, we first need to determine the electron configuration of Vanadium.. Recall that for a neutral element, Atomic number = # of protons = # of electrons. The atomic number of Vanadium is 23 and since it's a neutral element, this means V has 23 electrons.
What is the correct electron configuration for V^2+?. I'm studying for a placement exam for Chemistry ... Choose an expert and meet online.
Example of following the Aufbau principle, Pauli principle, and Hund's rule to construct an orbital diagram for a vanadium (Z=23) atom.
Choose the correct orbital diagram for vanadium [AR] 4s [||] [ { || } { | } { } { } { } ] ... metallic character decreases as you move to the right across a row in the periodic table and decreases as you move down a column d) metallic character increases as you move to the right across a row in the periodic table and decreases as you move down ...
Give the complete electronic configuration for Mn. Choose the statement that is TRUE. *Core electrons effectively shield outer electrons from nuclear charge. *Core electrons are the easiest of all electrons to remove. *Outer electrons efficiently shield one another from nuclear charge. *Valence electrons are most difficult of all electrons to ...
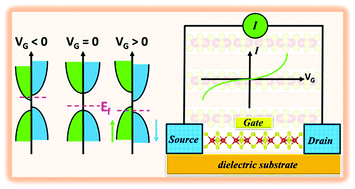
Doping Isolated One Dimensional Antiferromagnetic Semiconductor Vanadium Tetrasulfide Vs4 Nanowires With Carriers Induces Half Metallicity Journal Of Materials Chemistry C Rsc Publishing
Cr and Cu, as well as Cu and Ag, are exceptions in the "typical" filling order.Orbital Diagram. 1s Vanadium pentoxide (V2O5) is used as a catalyst, dye and color-fixer. Sources Found in the minerals patronite (VS4), vanadinite [Pb5 (VO4)3Cl], and carnotite [K2 (UO2)2 (VO4)H2O]. Pure metal produced by heating with C and Cl to produce VCl3 which is .

1 Write Orbital Diagrams For Each Of These Ions A V5 B Cr3 C Ni2 D Fe3 2 Determine If The Ion Is Diamagnetic Or Paramagnetic A V5 B Cr3 C Ni2

Which Of The Following Choices Is The Correct Electron Configuration For A Vanadium Atom 3d A Homeworklib
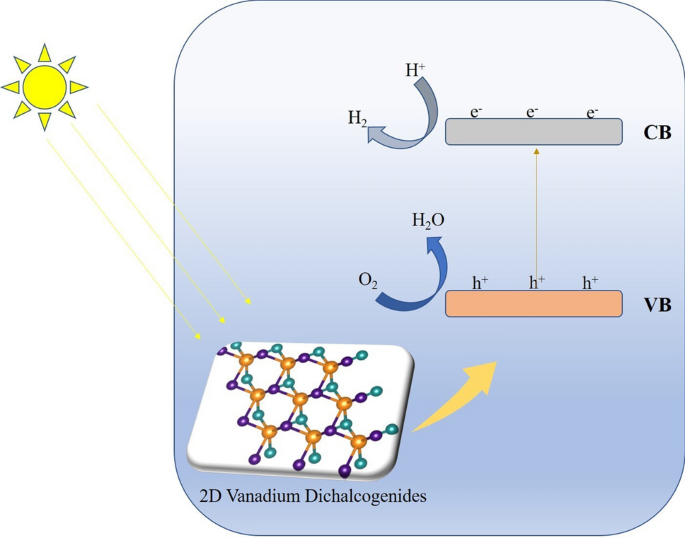
Pristine And Janus Monolayers Of Vanadium Dichalcogenides Potential Materials For Overall Water Splitting And Solar Energy Conversion Springerlink



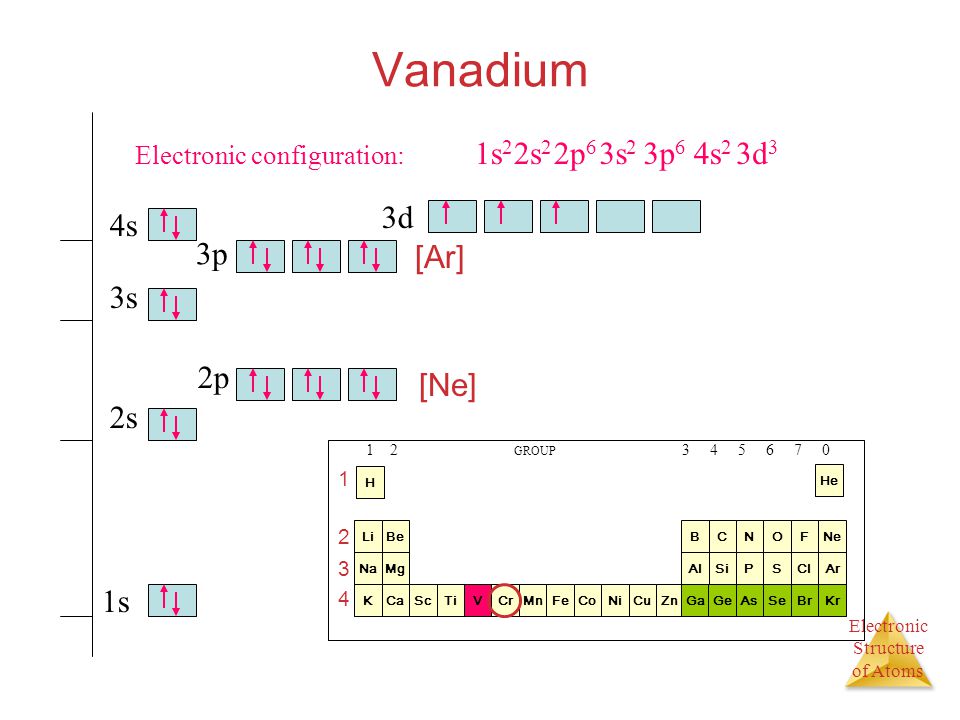




















0 Response to "44 choose the correct orbital diagram for vanadium."
Post a Comment