41 silicon electron dot diagram
A. Lewis electron dot diagram. (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. These dots are arranged to the right and left and above and below the ... Silicon tetrabromide is the inorganic compound with the formula SiBr 4. This colorless liquid has a suffocating odor due to its tendency to hydrolyze with release of hydrogen bromide. The general properties of silicon tetrabromide closely resemble those of the more commonly used silicon tetrachloride.
Silicon is in Group 14 (sometimes called Group IV or 4). Since it is in Group 4 it will have 4 valence electrons. When you draw the Lewis structure for Silicon ...2 answers · 1 vote: Apparently there are different schools of thought on this (How to Draw Electron Dot Diagrams ...
Silicon electron dot diagram
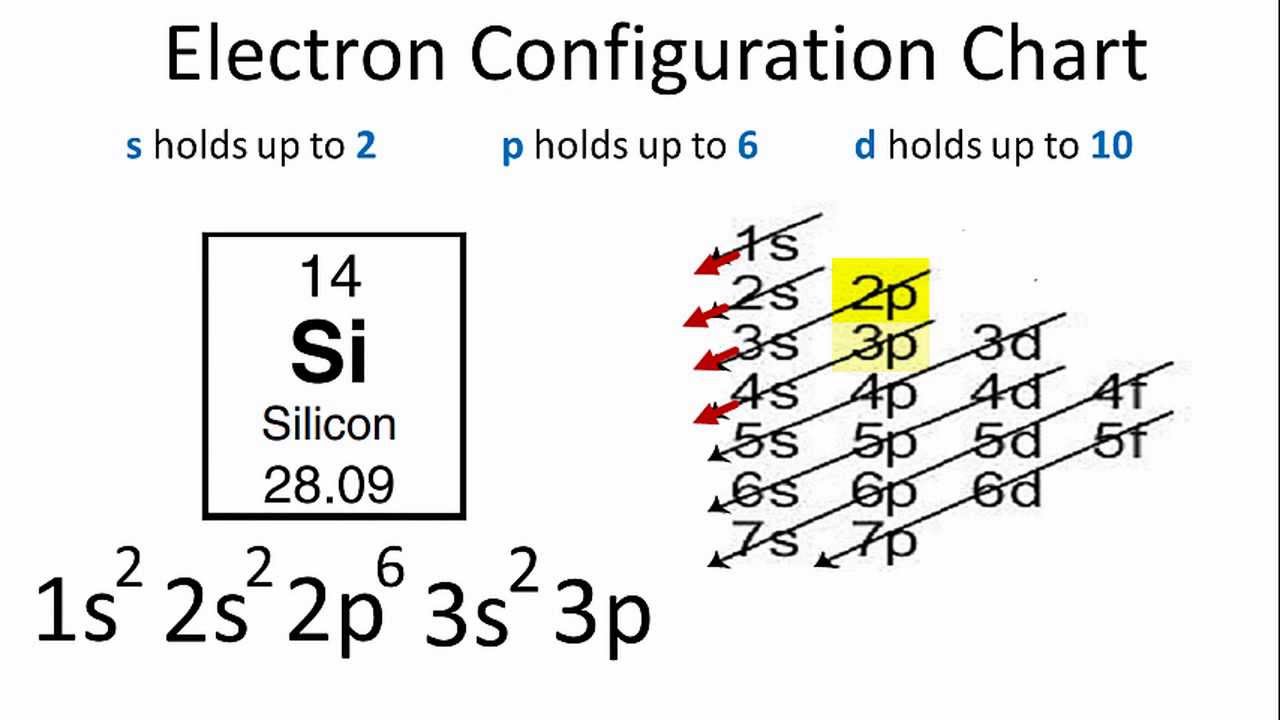
A step-by-step explanation of how to draw the SiH4 Lewis Dot Structure (Silicon Tetrahydride).For the SiH4 structure use the periodic table to find the total... Since silicon is in the 14th group of the periodic table and oxygen is in the 16th group, silicon has a 4 valence electron and oxygen has 6. Oxygen’s electronic configuration = 1s2 2s2 2p4 Silicon’s electronic configuration = 1s2 2s2 2p6 3s2 3p2 Hence, No. of valence electrons in oxygen = 6 No. of valence electrons in Silicon = 4 Silicon carbide | SiC - PubChem. National Center for Biotechnology Information. 8600 Rockville Pike, Bethesda, MD, 20894 USA. Contact. Policies. FOIA. National Library of Medicine. National Institutes of Health. Department of Health and Human Services.
Silicon electron dot diagram. Write the electron dot (Lewis) diagrams for the following. 9. carbon silicon Lewis electron dot diagrams for ions have less (for cations) or more (for anions) dots than the corresponding atom. Exercises Explain why the first two dots in a Lewis electron dot diagram are drawn on the same side of the atomic symbol. Silicon disulfide | S2Si - PubChem. National Center for Biotechnology Information. 8600 Rockville Pike, Bethesda, MD, 20894 USA. Contact. Policies. FOIA. National Library of Medicine. National Institutes of Health. Department of Health and Human Services. silicon In an electron dot diagram of potassium There is one dot. In an electron dot diagram of silicon there are four dots. Which element would you expect to be more reactive? silicon What is the... Silicon tetraiodide is the chemical compound with the formula Si I 4. It is a tetrahedral molecule with Si-I bond lengths of 2.432 (5) Å. SiI 4 is a precursor to silicon amides of the formula Si (NR 2) 4 (R = alkyl). It has also been of interest in the manufacture and etching of silicon in microelectronics .
Lewis Structure: A Lewis structure is a diagram that describes the covalent bonding between two or more atoms present in a molecule or ion. The atoms are written in terms of their elemental symbols. A step-by-step explanation of how to draw the Lewis dot structure for Si (Silicon). I show you where Silicon is on the periodic table and how to determine h... 0 people liked this ShowMe. Flag ShowMe. Viewed after searching for: cacl2 lewis structure. Lewis electron dot diagram for caBr. Lewis dot structure for Al2O3. Lewis electron dot diagram for MgCl2. lewis dot structure for covalent compounds. You must be logged into ShowMe. Electron-dot notation is used for drawing the lewis structure. In this, electrons are represented as dots around the symbol of the element. Steps to draw the lewis structure of SiBr4. Step 1: Count the number of valence shell electrons on each atom of the molecule to get the total valence electron count. SiBr4 has two elements ie; Si and Br.
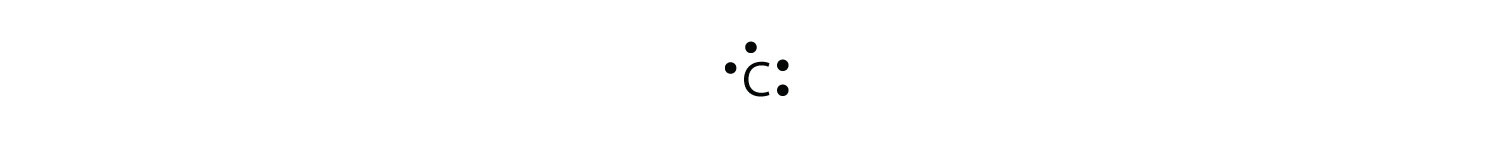
There are four valance electrons in Silicon, therefore there will be 4 dots in your electron dot diagram. And then the silicon is able to share in four bonds. Each of those bonds have two electrons, so the silicon is also feeling good about the octet rule. So I would feel very confident in this being the Lewis diagram, sometimes called the Lewis structure, for silicon tetrafluoride. silicon electron dot diagram. phosphorus electron dot diagram. sulfur electron dot diagram. calcium electron dot diagram. argon electron dot diagram. full outer shell. ... Start studying Electron Dot Diagrams. Learn vocabulary, terms, and more with flashcards, games, and other study tools. Search. Create. Electron Dot Diagrams. STUDY ... Let's do the Lewis structure for SiH4. On the periodic table, Silicon, group 4, 4 valence electrons; Hydrogen, group 1, 1 valence electron, but we have four of them. So 4 plus 4: 8 total valence electrons. Put the Si in the center, Hydrogens always go on the outside.
Electron dot diagram of a Silicon atom Electron dot diagram also called lewis structure which represents the valence electrons of atoms. As, from the Bohr diagram of Silicon, we got to know, it has only 4 valence electrons. So, just represent the 4 valence electrons around the Silicon atom as a dot. The electron configuration of Silicon
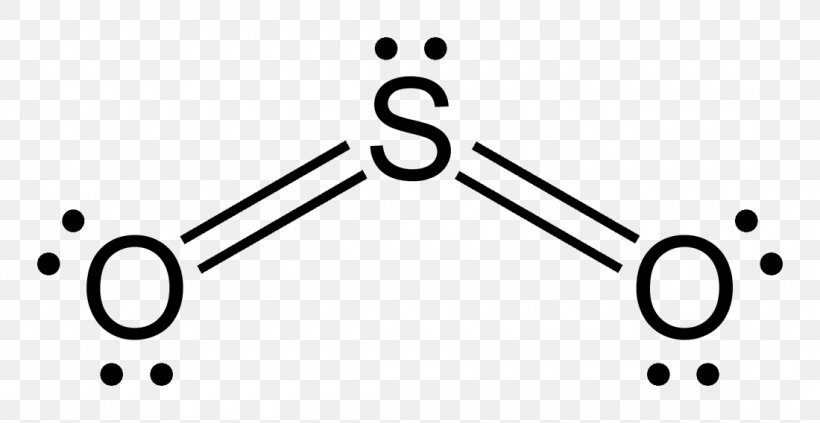
The Lewis structure for silicon disulfide is to be predicted. Concept introduction: The strategy for drawing Lewis structure is mention below. Calculate the number of valence electrons present in the molecule. Calculate the electron pairs by diving number of valence electrons by 2. Determine the bond pairs. Determine the lone pairs.
SiCl4 lewis's structure is straightforward and very easy to draw. "Lewis diagram describes the chemical bonding of atoms within a molecule". Lewis structure of SiCl4 contains 12 lone pairs on surrounding atoms and zero on the central atom. There are 4 bonding pairs present in the lewis structure of Silicon tetrachloride.
Answer (1 of 7): Silicon carbide (rarely: the mineral moissanite) is a refractory solid with a number of different allotropic covalent network structures. All of them have the atoms bound to four neighbors in a tetrahedral fashion with four covalent \sigma-bonds to the neighboring atom. One struc...
SiO2 Lewis Structure (Step by Step Construction) In the SiO 2 lewis Structure, the overall ratio of silicon to the oxygen atom is 1:2.The Silicon oxygen bonds are strong and keep the atoms firmly in place. Following are the steps to construct the SiO 2 Lewis Structure.. Step-1: Count the valence electrons of atoms For the SiO 2 Lewis structure, we need to figure out the number of valence ...
Silicon is in Group 14 (sometimes called Group IV or 4). Since it is in Group 4 it will have 4 valence electrons. When you draw the Lewis structure for Silicon you'll put four "dots" or valance electrons around the element symbol (Si). Hope this helped! 1.5K views View upvotes Promoted by HiddenWealth Where is Mark Cuban investing? Sophie S.
Silicon is in group 14 and period 3 of the periodic table and has four valence electrons in its Lewis structure. The four valence electrons means that silicon can bond in a way similar to carbon ...
molecule sih4 polar non lewis structure silicon draw tetrahydride . sih4 silane shape 2d lewis structure bond svg aldehyde pixels . sih4 geometry silane lewis structure silicon molecular electron tetrahydride dot draw hybridization tetrahedral chemistry polarity sih 3d sp known better .
Silicon is the second most abundant element and comprises 25% of the earth's crust. Makes up major portion of clay, granite, quartz (SiO 2 ), and sand. Approximate annual world production wide production: electronics grade pure silicon, 5000 tons; metallurgical grade, 480,000 tons; ferrosilicon, 3,400,000.
On the periodic table, Si is in group 4, it has 4 valence electrons. Oxygen has 6, but we have two Oxygens, for a total of 16 valence electrons. We'll put the Si in the center and then the Oxygens on either side. We'll put two electrons between atoms to form bonds, and the rest around the outside atoms. Two, 4, 6, 8, 10, 12, 14, 16.
Check the Formal Charges to make sure you have the best Lewis Structure. Explain How Examples: SO 4 2-, N 2 O, XeO 3; Notable Exceptions to the Octet Rule. H only needs 2 valence electrons. Be and B don't need 8 valence electrons. S and P sometimes have more than 8 val. Electrons.
Solution. Having lost its two original valence electrons, the Lewis electron dot diagram is just Ca 2+. Ca2+. The O 2− ion has gained two electrons in its valence shell, so its Lewis electron dot diagram is as follows: Test Yourself. The valence electron configuration of thallium, whose symbol is Tl, is 6 s2 5 d10 6 p1.
Silicon carbide | SiC - PubChem. National Center for Biotechnology Information. 8600 Rockville Pike, Bethesda, MD, 20894 USA. Contact. Policies. FOIA. National Library of Medicine. National Institutes of Health. Department of Health and Human Services.
Since silicon is in the 14th group of the periodic table and oxygen is in the 16th group, silicon has a 4 valence electron and oxygen has 6. Oxygen’s electronic configuration = 1s2 2s2 2p4 Silicon’s electronic configuration = 1s2 2s2 2p6 3s2 3p2 Hence, No. of valence electrons in oxygen = 6 No. of valence electrons in Silicon = 4
A step-by-step explanation of how to draw the SiH4 Lewis Dot Structure (Silicon Tetrahydride).For the SiH4 structure use the periodic table to find the total...





















0 Response to "41 silicon electron dot diagram"
Post a Comment